金桔
金币
威望
贡献
回帖0
精华
在线时间 小时
|
登陆有奖并可浏览互动!
您需要 登录 才可以下载或查看,没有账号?立即注册


×
Western blot是分子生物学,蛋白表达及组学和分子克隆实验必须掌握的实验技术。概念性的东西不再详述,一些决定成败的细节才是我写这篇文章的目的。
Western blot流程
A提蛋白---B电泳---C Transblot---D抗原抗体反应---E检测。
需要注意的细节(乱序版)
1. 裂解蛋白冰上操作。
2. 蛋白裂解需要根据实验目的和蛋白的定位选择合适的细胞裂解液。如核蛋白有专门的提取试剂盒。
3. 蛋白裂解需要加入磷酸酶抑制剂,尤其是做磷酸化蛋白。
4. 蛋白定量时注意浓度不要超出标曲检测线,线性要达到2个9。
5. 蛋白热变性过程中防止EP管爆开。
6. PVDF膜通常有两种孔径0.45和0.22μM(小于20kDa)。
7. 根据蛋白大小选择不同浓度的分离胶,有商品化的预制胶出售。
8. 转膜做三明治时一定注意排除气泡。
9. 条带定量分析软件imageJ下载网址:https://http://imagej.net
10. 什么是抗体。
抗体,也叫免疫球蛋白(Ig),是一种特异性结合抗原的糖蛋白,而抗原是在易感染动物体内引发抗体产生的物质。在体内,抗体是由于外源性分子的侵袭而产生的。抗体以一个或者多个Y字形单体存在,每个Y字形抗体由4条多肽链组成,包含两条相同的重链和两条相同的轻链。轻链和重链是根据它们分子量大小命名的。Y子形结构的顶端是可变区,为抗原结合部位。
11. Western blot,ELISA和免疫组织化学法均是检测蛋白表达水平,不是所有的蛋白都适合western blot技术去检测。
12. 一抗来源和种属尽量选择不同的。
13. 磷酸化抗体使用5%BSA封闭,勿使用牛奶。
14. 内参蛋白的选择
包浆蛋白和全细胞蛋白常使用β-actin,GAPDH,β-Tubulin。
15. 玻璃板洗干净。
16. 显影条带分子质量与marker不完全匹配
可能原因有目标蛋白的翻译后修饰,剪切,多聚体形成相关。
17. 背景脏
抗体浓度过高,封闭时间过短,洗涤不充分,尽量不要多张膜一起洗涤。
18. 抗体的价格一般会比官网的便宜一些,经常会有买一送一或打折的活动。
19. PMSF,全称为Phenylmethanesulfonyl fluoride,苯甲基磺酰氟,是一种广泛性蛋白酶抑制剂。
20. 使用电动研磨仪研磨组织时注意过程会产热,注意冰上操作防止蛋白降解。
21. RIPA和PMSF现用现配。
22. 甲醇的目的主要有防止凝胶溶胀,降低蛋白转移速度,使蛋白与SDS解离。
23. 加入SDS的目的:使蛋白带上负电荷,使蛋白快速穿过膜,减少蛋白沉淀,减弱蛋白与膜的吸附。
24. 1xtrans buffer(10xtrans buffer 80ml+甲醇160ml+双蒸水560ml)
25. 封闭液配置:称取1gBSA,加入20mlTBST配成5%,混匀。
26. 膜再生液可以对同一张膜进行多次western blot检测。
27. 显影结果没有条带的原因:一抗二抗不匹配;蛋白上样量太少,尤其是核蛋白提的是总蛋白的情况;一抗效价太低;组织样本放置时间过长降解;转膜没有转上或转过;显影液失效或显影时间过短。
28. 杂带太多解决方案:使用单克隆抗体;降低一抗浓度;降低蛋白上样量。
29. 条带上有圆形白孔:转膜的时候有气泡;孵育抗体接触不良。
30. 缺带或带显不全的原因:表达丰度低,多张膜共同孵育。
31. 膜上有黑色颗粒状杂质:分泌液未完全溶解有杂质;增多洗膜次数。
32. 高背景:二抗浓度过高;封闭不充分;
33. 笑脸条带:迁移过快;电泳温度过高;凝胶未完全凝固。
34. 条带拖尾:分离胶浓度过大;样品降解;电泳液过度使用。
35. 牵手条带:分离胶选择不合适,上样量过大。
36. 条带不在一条直线上:分离胶不均匀。
37. Western blot为造假重灾区,一定要备份原始数据,原始图片。
38. 玻璃板与梳子有1.0mm/1.5mm,梳子有10孔和15孔,注意统一。
39. 电泳:先选择80V恒压电泳,待溴酚兰迁移至分离胶后(约半小时),提高电压至100-120V继续电泳,总时间一般2-3h,电泳至溴酚兰快跑出即可终止电泳,进行转膜。
40. 转一张膜需准备2张10×10cm海绵、4张7.0×8.0cm的滤纸和1张7.5×8.5cm的PVDF膜。将切好的PVDF膜先置于100%甲醇中浸湿15s,然后移至超纯水中浸泡2min,最后转至转移液中至少平衡5min方可使用。
41. 电泳完毕后切下来的分离胶直接进入转移液中平衡15-30min,以除去SDS对蛋白转移的影响。
42. 小心从转移液中取出平衡好的分离胶盖于滤纸上(若是普通湿转,胶反面朝上,则转膜后PVDF膜蛋白面与上样一致。膜两边的滤纸不能相互接触,接触后会发生短路影响蛋白的转移。
43. 据蛋白分子量的大小选择不同的转膜时间,一般70KDa以内的蛋白可以用200mA电转1.5h,20KDa以内的小蛋白建议75V条件下电转40min。
44. 关于Western blot投稿自查注意事项:过度曝光;模糊;选择合适的内参;防止内参重复使用;原始数据。
45. TEMED添加过量会使胶凝得过快并且孔径大小不一,影响蛋白的电泳,甚至可能烧胶。
46. 电转液中一般需加入甲醇或乙醇,而电转时的放热会加速醇类的挥发进而影响下一次重复使用时的电转效果。重复使用电转液时应考虑到醇类的挥发而适当延长电转时间,最好是每次电转时都使用新的电转液。
47. 转移大于100KDa的蛋白时,需要在转膜液中加入适量SDS。
48. 取膜时,使用镊子夹取膜边缘,避免膜被杂蛋白和油脂污染。
49. 任何时候都避免干膜。
SCI描述western blot实验范文
1.PMID:30296700
Methods
Western blot analysis. Western blot was performed as previously described. Rats were anesthetized with isoflurane at 24h after SAH and transcardially perused with 100 ml of chilled PBS (0.01 M, pH7.4) before brains collection to ensure that there were no blood contaminants. The left hemispheres were instantly collected and snap frozen in liquid nitrogen, then placed in -80℃ for storage until use. Brain samples were homogenized in RIPA lysis buffer ( sc-24948, Santa Cruz Biotechnology Inc., TX, USA) and further centrifuged at 14,000g at 4℃ for 30 min. Equal amounts of protein (30 μg) were loaded onto 7.5% − 12.5% SDS-PAGE gel, then electrophoresed and transferred to nitrocellulose membranes (0.2 µm), which were blocked with 5% non-fat blocking grade milk (Bio-Rad, Hercules, CA, USA) and incubated with the following primary antibodies overnight at 4 ℃: anti-Mas (1:2000, NBP178444, NOVUS Biologicals, CO, USA), anti-PKA-Cα (1:3000, 4782, Cell Signaling Technology Inc., MA, USA), anti-p-CREB (1:1000, 9198, Cell Signaling Technology Inc., MA, USA), anti-CREB (1:1000, 9197, Cell Signaling Technology Inc., MA, USA), anti-UCP-2 (1:2000, 89326, Cell Signaling Technology Inc., MA, USA), anti-Bcl-2 (1:2000, ab59348, Abcam, MA, USA), anti-Bax (1:4000, ab182734, Abcam, MA, USA), anti-Romo-1 (1:200, AVIVA Systems Biology, CA, USA), and anti-β-actin (1:4000, sc47778, Santa Cruz Biotechnology Inc., TX, USA). On the following day, the membranes were incubated with the appropriate secondary antibody (1:4000, Santa Cruz Biotechnology Inc., TX, USA) at room temperature for 1 h. Immunoblots were then visualized with ECL Plus chemiluminescence reagent kit (Amersham Bioscience, PA, USA) and quantified with optical methods using the ImageJ software (ImageJ 1.5, NIH, USA). The results were normalized using β-actin as an internal control.
Figure legend
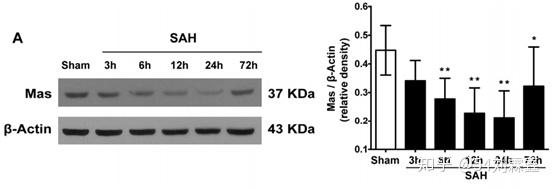
Representative Western blot image and quantitative analysis of Mas from the ipsilateral hemisphere in time course experiment after SAH. Data represent with mean ± SD (n = 6 per group). *P < 0.05, * *P < 0.01 vs. Sham group.
Results
Western blot results showed that expression of Mas dramatically decreased after SAH induction and reached to the lowest level at 24 h when compared to Sham group.
2.PMID: 30703614
Methods Western blot
Western blot analysis was performed as previously described. After sample preparation, equal amounts of a sample protein (50 μg) were loaded onto an SDS-PAGE gel. After electrophoresis, the samples were transferred onto a nitrocellulose membrane. Then, the membrane was blocked for 2 h at room temperature and incubated overnight at 4 °C with the following primary antibodies: anti-apoE (1:500, Abcam, Cambridge, MA, USA), anti-Iba-1 (1:1000, Wako, USA), anti-APP (1:1500, Abcam, Cambridge, MA, USA), anti-myelin basic protein (MBP, 1:1000, Abcam, Cambridge, MA, USA), anti-LRP1 (1:1000, Abcam, Cambridge, MA, USA), anti-Shc1 (1:2000, Abcam, Cambridge, MA, USA), anti-PI3K (1:1000, Cell signaling, USA), anti-Akt (1:1000, Cell signaling, Danvers, MA, USA), anti-phospho-Akt (p-Akt, 1:1000, Cell signaling, Danvers, MA, USA), anti-CD16 (1:1500, Santa Cruz, Dallas, TX, USA), anti-iNOS (1:500, Abcam, Cambridge, MA, USA), anti-CD206 (1:1500, Santa Cruz, Dallas, TX, USA), and anti-β-actin (1:5000, Santa Cruz, Dallas, TX, USA). Appropriate secondary antibodies (1:5000, Santa Cruz, Dallas, TX, USA) were selected to incubate with the membrane for 2 h at room temperature. Then, blot bands were visualized with an ECL reagent (Amersham Biosciences UK Ltd., PA, USA). Non-saturated bands were selected to perform densitometry quantification using Image J software (Image J 1.51, NIH, USA).
Figure legend
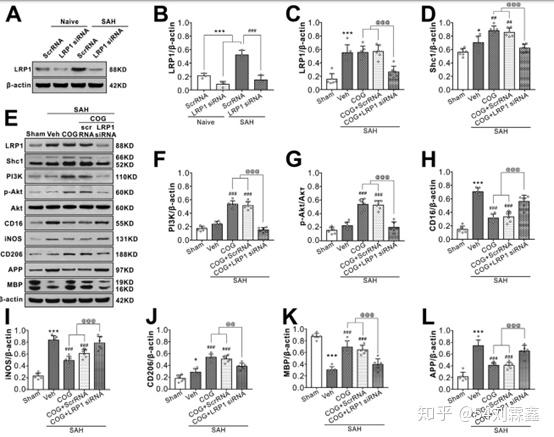
LRP1-knockout abolished the beneficial effect of COG1410 on M2 microglia polarization, white matter protection at 24 h after SAH. (AG, I–K) Representative western blot bands and quantification of LRP1, Shc1, PI3K, p-Akt/Akt, CD16, iNOS, CD206, APP, and MBP. Data was represented as mean ± SD (n = 6 per group). (H) LRP1 was binding to endogenous apoE and Shc1 in brain tissue from Sham or SAH rats. One-way ANOVA was used followed by Tukey’s HSD post hoc test and Holm-Bonferroni correction. *P < 0.05, ***P < 0.001 vs. Sham group; ###P < 0.001 vs. SAH + Vehicle group; @P < 0.05, @@P < 0.01, @@@P < 0.001 vs. SAH + COG + LY294002 group. Vehicle, sterile 0.9% of NaCl; COG, COG1410; DMSO, Dimethyl sulfoxide, LY294002, PI3K inhibitor.
Results
Injection of LRP1 siRNA via i.c.v 48 h before SAH induction significantly decreased the expression of LRP1 both in naïve and SAH rats, which confirmed the knockdown efficacy of LRP1 siRNA in the present study (Fig. 8A, B). Although COG1410 treatment had no effect on in LRP1 expression after SAH, it significantly increased the protein levels of Shc1, PI3K, phosphorylated-Akt (Ser473), microglia M2 phenotype marker CD206 and myelin marker MAP but decreased protein levels of M1 phenotype marker CD 16 and iNOS, axonal injury marker APP when compared with vehicle treated SAH rats. When LRP1 expression was knockdown in the SAH + COG1410 + LRP1 siRNA group, the effects of COG1410 on Shc1/PI3K/Akt signaling pathway activation, M2 microglial polarization and white matter protection were reversed when compared with the SAH + COG1410 + Scr siRNA group (Fig. 8C–L).
3. PMID: 31160314
Methods
Western Blot Analysis. Membrane proteins from rat liver samples (from the median liver lobe) were extracted using a proteoExtract native membrane protein extraction kit (catalog number 444810; Calbiochem, San Diego, CA). The total protein was determined by a Pierce BCA Protein Assay Kit (Thermo Fisher Scientific) using the manufacturer protocol. . Samples for Western blots were prepared according to the manufacturer instructions (Thermo Fisher Scientific); 35 mg was loaded onto Bis-Tris 4%–12% (Oatp1a1, Oatp1a4, Oatp2b1, Ostb, Mdr1) or Tris Acetate 7% (Mrp2, Mrp3) gels. After electrophoresis, the transfer of proteins to polyvinylidene fluoride membranes according to standard procedures was performed. Each protein was analyzed separately on a different membrane, and ATPase was probed on the same blot as the respective protein and used as the loading control. The membranes were incubated with antibody anti-Oatp1 (1:200; catalog number AB3670P; Millipore, Burlington, MA), anti-Oatp1a4 (1:500; catalog number AB3672P; Millipore), anti-Oatp1b2 (1:100; catalog number sc-376904; Santa Cruz Biotechnology, Dallas, TX), anti-Mrp3 (1:1000; catalog number 399095; Cell Signaling Technology, Danvers, MA), anti-Ostb antibody (1:150; catalog number HPA008533; Sigma-Aldrich), anti-Mrp2 (1:20; catalog number MC-206; Kamiya Biochemical Company, Seattle, WA), anti-Mdr1b (1:100; catalog number sc-55510; Santa Cruz Biotechnology), or anti- ATPase (1:5000; catalog number sc-28800; Santa Cruz Biotechnology) overnight in diluted 5% (w/v) bovine serum albumin-Tris–buffered saline Tween 20. After washing, the blots were probed with horseradish peroxidase–conjugated anti-rabbit (for Oatp1a1, Oatp1a4, Ostb, and Mrp3; 1:7000; catalog number sc-2030; Santa Cruz Biotechnology) or horseradish peroxidase–conjugated anti-mouse IgG secondary antibody (for Oatp1b2, Mdr1, and Na1/K1 ATPase; 1:10,000; catalog number 115-035-003; Jackson ImmunoResearch, West Grove, PA) in 5% milk for 1 hour at room temperature. Detection was enhanced using chemiluminescence reagents (ECLTM Select; GE Healthcare Bio-Sciences, Piscataway, NJ) and measured with ImageLab software version 4.1 (Bio-Rad, Hercules, CA). . The data were normalized to loading control (ATPase) using densitometric analysis to compare protein expression between WT and PCK rats with ImageJ version 1.51 (National Institutes of Health).
Figure legend
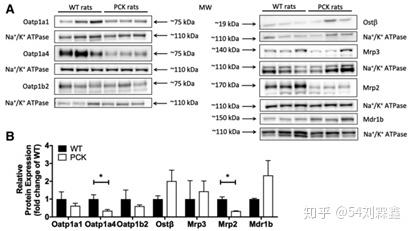
Transporter protein expression in WT compared with PCK rat livers. (A) Immunoblots of liver tissue samples from WT and PCK rats (n 5 3/group). Each protein was analyzed on a separate membrane, and Na1/K1 ATPase was probed on the same blot as the respective protein. (B) Results of integrated optical density analysis of Western blots prepared from WT and PCK rat liver tissue samples (n 5 3/group). Data were normalized to Na1/K1 ATPase and are shown as the mean 6 S.D. Statistically significant differences were determined by an unpaired, two-tailed t test.
Results
Protein expression levels were determined using Western blot analysis (Fig. 3) and LC-MS/MS proteomic analysis (Fig. 4). Immunoblot analysis of liver tissue from WT and PCK rats revealed a significant decrease in Mrp2 and Oatp1a4 (3.0-fold and 2.9-fold, respectively) (Fig. 3) in PCK rat livers. A 1.4-fold, 2.0-fold, and 2.3-fold increase in protein levels were detected in PCK rat livers for Mrp3, Ostb, and Mdr1b, respectively, but these differences were not statistically significant (Fig. 3). In addition, protein levels of Oatp1a1 and Oatp1b2 were decreased by 1.6-fold and 1.7-fold, respectively, in PCK rat livers, but differences were not significant (Fig. 3).
4.PMID:32381096
Methods
Western blot analysis.
Western blot analysis was performed as previously reported. Proteins of the ipsilateral hemisphere were extracted by homogenizing in radio-immunoprecipita tion assay lysis buffer. Equal amounts of a sample protein were loaded onto an SDS-PAGE gel. First, electrophoresis and transfer of the samples to a nitrocellulose membrane were performed. Second, the membrane was blocked for 2 h at room temperature and incubated overnight at 4 °C with the following primary antibodies: anti-TGR5 (1:1000, Abcam), anti-BRCA1 (1:1000, Santa Cruz Biotechnology), anti-Sirt1(1:1000, Abcam), antioccludin (1:2000, Abcam, USA), anti-ZO-1(1:200, Santa Cruz Biotechnology) and anti-β-actin (1:5000, Santa Cruz Biotechnology). The secondary antibodies were all from Santa Cruz Biotechnology. Blot bands were visualized with an ECL reagent (Amersham Biosciences UK Ltd., PA, USA) and were quantified by densitometry using Image J software (Image J 1.4, NIH, USA).
Figure legend
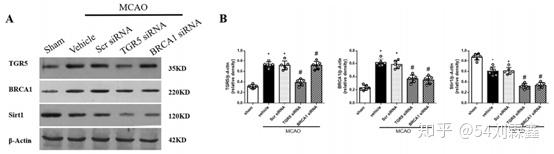
The effect of knockdown TGR5 on BRCA1/Sirt1 expression and BBB permeability after MCAO. a The band of Western blot analysis; b The relative density of TGR5, BRCA1, Sirt1.n = 6 per group.
Results
The results of Western blot staining showed that TGR5 expression was partially prevented by TGR5 siRNA (Fig. 6a and b). Compared with scramble siRNA group, TGR5 siRNA significantly inhibited expressions of BRCA1 and Sirt1 after MCAO (P < 0.05) (Fig. 6a and b). The knockdown efficacy of BRCA1 siRNA was also confirmed by Western blot and BRCA1 knockdown markedly decreased the Sirt1 expression and had no effect on TGR5 expression after MCAO (Fig. 6a and b).
5.PMID:27995618
Methods
Western blot analyses Whole-cell lysates were prepared for Western blotting. Twenty-five micrograms of cell protein was electrophoretically separated on a 10% SDS–polyacrylamide gel and transferred to a PVDF membrane,which was then blocked by incubation for 1 hr at room temperature in 5% fat-free dry milk in Tris-buffered saline containing 01% Tween 20 (TBS-T). The blots were then incubated overnight at 4° with rabbit antibodies against mouse GAPDH (sc-20357, Santa Cruz Biotech) and Foxd3 (ab107248, abcam corp., Cambridge, MA) diluted 1 : 1000 in TBS-T containing 5% bovine serum albumin, washed for 25 min with TBS-T, and incubated for 1 hr at room temperature with horseradish peroxidase-conjugated secondary F(ab’)2 (Zymed Laboratories, San Francisco, CA) (1 : 20 000 in TBS-T containing 5% bovine serum albumin), then bound antibody was visualized using the ECL detection system (Amersham, Arlington Heights, IL).
原文地址:https://zhuanlan.zhihu.com/p/309735533 |
|
 /3
/3 